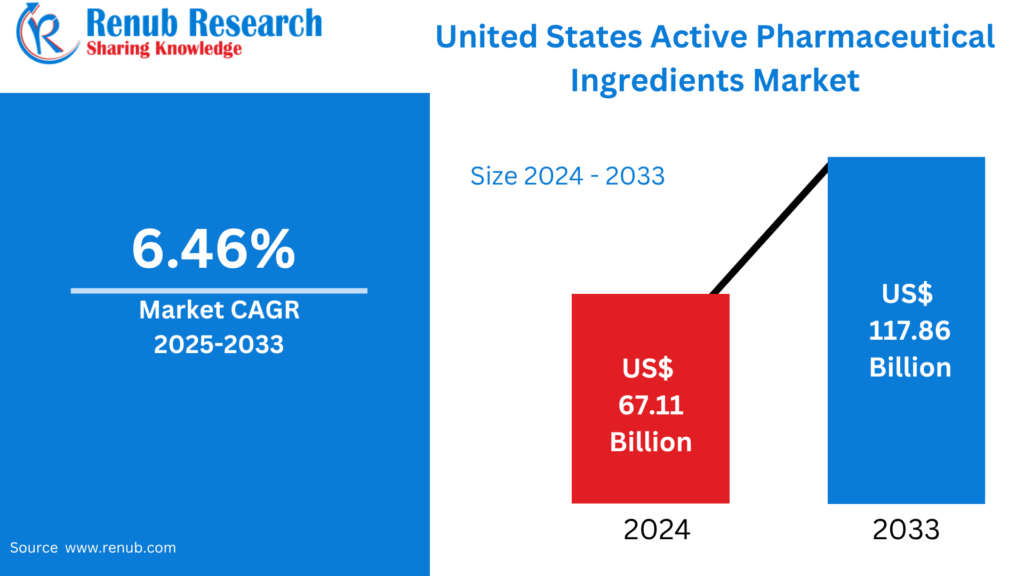
United States Active Pharmaceutical Ingredients Market Size and Forecast (2025–2033)
According to Renub Research United States Active Pharmaceutical Ingredients (API) Market is positioned for sustained growth over the forecast period, driven by strong pharmaceutical demand, innovation in drug development, and rising healthcare needs. The market was valued at US$ 67.11 billion in 2024 and is projected to reach US$ 117.86 billion by 2033, expanding at a compound annual growth rate (CAGR) of 6.46% from 2025 to 2033.
This growth is underpinned by increasing consumption of both branded and generic drugs, the growing prevalence of chronic and lifestyle-related diseases, and rapid advancements in biopharmaceutical and biotechnology-based therapies. The United States remains one of the most influential pharmaceutical markets globally, supported by strong regulatory oversight, advanced research infrastructure, and continuous innovation across the drug value chain.
Request a free sample copy of the report:https://www.renub.com/request-sample-page.php?gturl=us-active-pharmaceutical-ingredients-market-p.php
United States Active Pharmaceutical Ingredients Market Overview
Active Pharmaceutical Ingredients (APIs) are the core biologically active substances in pharmaceutical products that produce the intended therapeutic effect. APIs are manufactured through chemical synthesis, fermentation, or biotechnological processes and are subsequently formulated with excipients to produce finished dosage forms such as tablets, capsules, injectables, and biologics.
In the United States, APIs form the backbone of the pharmaceutical ecosystem, enabling the production of a wide range of medicines, from small-molecule generics to complex biologics. The U.S. pharmaceutical industry has evolved rapidly in response to growing healthcare demand, an aging population, and the increasing burden of chronic diseases such as diabetes, cardiovascular disorders, cancer, and neurological conditions.
The U.S. Food and Drug Administration (FDA) enforces stringent quality, safety, and compliance standards for API manufacturing. These regulations, while demanding, ensure product consistency and patient safety, strengthening confidence across healthcare providers and consumers. With robust domestic innovation, expanding drug pipelines, and increasing focus on supply chain resilience, APIs continue to be a strategic and indispensable segment of the U.S. pharmaceutical market.
Growth Drivers in the United States Active Pharmaceutical Ingredients Market
Increasing Prevalence of Chronic Diseases
The rising prevalence of chronic diseases is one of the most significant drivers of API demand in the United States. Conditions such as diabetes, cardiovascular disease, cancer, and chronic respiratory disorders require long-term or lifelong medication, directly increasing the volume and diversity of APIs consumed.
Lifestyle factors, including physical inactivity, poor nutrition, smoking, and alcohol consumption, combined with an aging population, are intensifying disease incidence. As a result, pharmaceutical companies are investing heavily in research and development to create effective, long-term therapeutic solutions. This trend drives continuous demand for high-quality APIs, particularly those used in maintenance therapies and combination drugs. Preventive care initiatives and early diagnosis are also expanding treatment populations, reinforcing sustained API consumption.
Expansion of the Generic Drug Market
The expiration of patents for several blockbuster drugs has opened substantial opportunities for generic drug manufacturers in the United States. Generic medicines rely on the same APIs as branded drugs but are offered at significantly lower prices, improving affordability and access to healthcare.
The U.S. FDA actively supports the approval of generics and biosimilars through accelerated review pathways, encouraging market competition and cost containment. As healthcare providers, insurers, and government programs prioritize cost-effective treatment options, demand for APIs used in generic formulations continues to rise. This trend has positioned generic APIs as a major growth pillar within the U.S. API market.
Technological Advancements in Biotech APIs
Technological innovation in biotechnology is reshaping the U.S. API landscape. Biotech APIs, produced using living systems such as cell cultures and recombinant DNA technology, offer targeted therapies with improved efficacy and reduced side effects compared to traditional chemical APIs.
Biologic drugs are increasingly used in oncology, autoimmune diseases, and rare disorders, driving demand for advanced API manufacturing capabilities. U.S.-based pharmaceutical and biotechnology companies are investing in state-of-the-art facilities for monoclonal antibodies, vaccines, and cell- and gene-based therapies. This transition toward biologics represents a critical growth phase for the U.S. API market, enhancing innovation, specialization, and domestic manufacturing capacity.
Challenges in the United States Active Pharmaceutical Ingredients Market
Supply Chain Disruptions and Import Dependence
Despite its strong pharmaceutical base, the United States remains partially dependent on API imports from countries such as India and China. This reliance exposes the market to supply chain disruptions caused by geopolitical tensions, trade restrictions, pandemics, and raw material shortages.
Such disruptions can delay drug manufacturing, affect availability, and increase costs across the healthcare system. In response, policymakers and industry stakeholders are emphasizing domestic API production and supply chain diversification to reduce vulnerabilities. While reshoring initiatives are gaining momentum, building long-term manufacturing resilience remains a complex and capital-intensive challenge.
Stringent Regulatory and Compliance Requirements
Regulatory compliance is both a strength and a challenge for the U.S. API market. The FDA’s rigorous standards for Good Manufacturing Practices (GMP), quality assurance, and documentation ensure high safety and efficacy levels. However, compliance requires substantial investment in infrastructure, skilled labor, and continuous audits.
These requirements can increase production costs and extend time-to-market, particularly for small and mid-sized manufacturers. While large pharmaceutical companies can absorb these costs, smaller players may face barriers to entry or expansion, limiting overall market competition.
United States Captive Active Pharmaceutical Ingredients Market
Captive API manufacturing refers to APIs produced in-house by pharmaceutical companies for their own drug development and production needs. In the United States, large pharmaceutical firms increasingly favor captive production to ensure quality control, protect intellectual property, and reduce dependence on external suppliers.
This model enhances supply chain reliability and allows companies to align API production closely with clinical trials and commercial demand. Captive manufacturing is particularly important for high-value, high-potency, and innovative APIs, where confidentiality and process optimization are critical.
United States Merchant Active Pharmaceutical Ingredients Market
Merchant API manufacturers produce APIs for sale to third-party pharmaceutical companies. This segment plays a vital role in supporting small and medium-sized drug manufacturers that lack in-house production facilities.
Merchant API suppliers offer scalability, technical expertise, and cost efficiency, enabling pharmaceutical companies to focus on formulation, branding, and distribution. The growing complexity of APIs and demand for specialized manufacturing have strengthened the role of contract development and manufacturing organizations (CDMOs) in the U.S. market.
United States Biotech Active Pharmaceutical Ingredients Market
The biotech API segment is experiencing rapid expansion due to the success of biologic medicines in treating complex diseases. These APIs are produced using advanced biotechnological processes and are central to therapies for cancer, autoimmune disorders, and rare diseases.
Government support, faster regulatory approvals, and increasing healthcare expenditure are encouraging investment in biotech API manufacturing. As biologics pipelines continue to grow, biotech APIs are expected to account for an increasing share of total API demand in the United States.
United States Synthetic Active Pharmaceutical Ingredients Market
Synthetic APIs, produced through chemical synthesis, remain the foundation of many conventional pharmaceuticals. They are widely used in treatments for cardiovascular, neurological, infectious, and metabolic diseases.
Despite growing competition from biologics, synthetic APIs continue to dominate due to their cost-effectiveness, scalability, and broad therapeutic application. Manufacturers are focusing on sustainable chemistry, high-purity production, and process optimization to meet regulatory and environmental requirements.
Therapeutic Application-Based Market Insights
Cardiovascular APIs represent a major segment due to the high prevalence of heart disease in the U.S. Oncology APIs are witnessing the fastest growth, driven by rising cancer incidence and demand for precision medicine. Orthopedic, nephrology, and neurology APIs are also expanding steadily, supported by aging demographics and increased awareness of chronic conditions.
Competitive Landscape and Key Players
The U.S. API market is highly competitive, with the presence of global pharmaceutical leaders and specialized manufacturers. Key players include Pfizer Inc., Novartis AG, BASF SE, Teva Pharmaceutical Industries Ltd, Viatris Inc., Sanofi Inc., Merck KGaA, Dr. Reddy’s Laboratories Ltd, Lupin Ltd, and Bristol-Myers Squibb.
These companies compete through innovation, capacity expansion, strategic acquisitions, and strong compliance capabilities.
Conclusion
The United States Active Pharmaceutical Ingredients market is set for robust growth from 2025 to 2033, driven by rising chronic disease burden, generic drug expansion, and rapid advancements in biotech and biologic therapies. While challenges such as supply chain dependence and regulatory complexity persist, strategic investments in domestic manufacturing, innovation, and compliance are strengthening the market’s long-term outlook. As the U.S. healthcare system continues to evolve, APIs will remain a critical pillar supporting pharmaceutical innovation, affordability, and patient care.